Exfoliation of Cu-Containing Poly(triazine imide): From Three-Dimensional to Two-Dimensional Particle Morphology
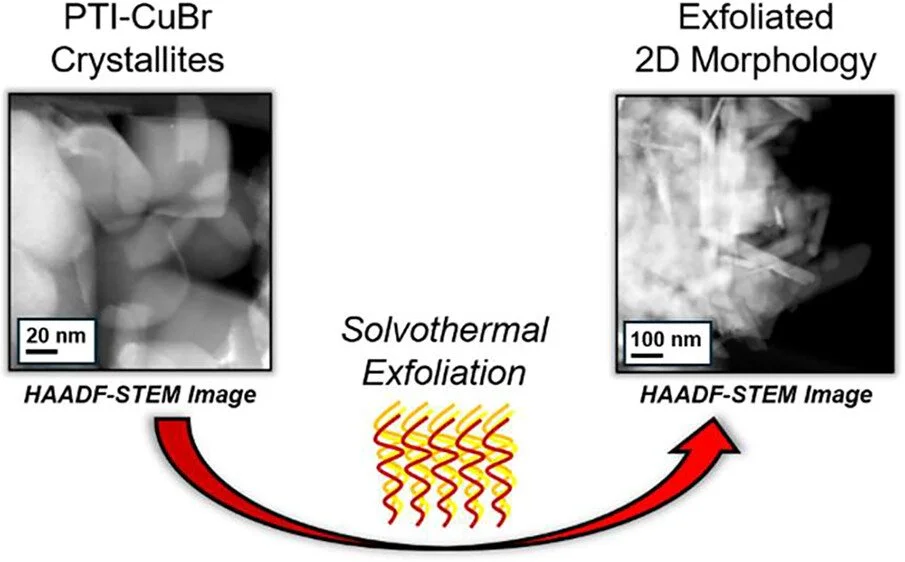
Controlling the morphological parameters of extended covalent organic frameworks remains challenging and represents an important yet often elusive metric of consideration. Typically, carbon nitride materials possess local ordering but remain largely amorphous in terms of their long-range order and orientation. This study probes the synthesis of a crystalline carbon nitride, poly(triazine imide) lithium bromide which possesses an atomically-precise extended structure, and demonstrates its exfoliation into a two-dimensional hexagonal sheet-like morphology. Furthermore, a previously unreported carbon nitride material, poly(triazine imide) copper bromide, or PTI-CuBr, was developed through an additional flux-assisted cation-exchange process and is shown to retain its internal Cu cations during solvothermal exfoliation. Characterization by dynamic light scattering and high-angle annular dark-field scanning electron microscopy reveals the morphological changes and captures the high aspect ratio of the thin carbon nitride sheets with <10 nm thickness while maintaining hundreds of nm in width. Additional characterization by energy-dispersive spectroscopy and X-ray photoelectron spectroscopy confirms that the Cu:Br:N molar ratio was maintained within the extended layers throughout the exfoliation process. This top-down synthesis approach differs from typical methods that isolate thin sheets for subsequent metal–cation coordination and illustrates the importance of maintaining oxygen-free conditions to minimize copper clustering. Thus, this new approach is demonstrated to provide a consistent and more homogeneous occupancy of the PTI pore spaces throughout the carbon nitride framework.
(1)McGuigan, S.; Ortiz, E. O.; Jeon, S.; Donley, C. L.; Stach, E. A.; Maggard, P. A. Exfoliation of Cu-Containing Poly(Triazine Imide): From Three-Dimensional to Two-Dimensional Particle Morphology. Langmuir 2026, 42 (6), 4489–4496. https://doi.org/10.1021/acs.langmuir.5c05167