Quantitative Analysis of the Semiconductor–Electrolyte Interface Using Cyclic Voltammetry Measurements
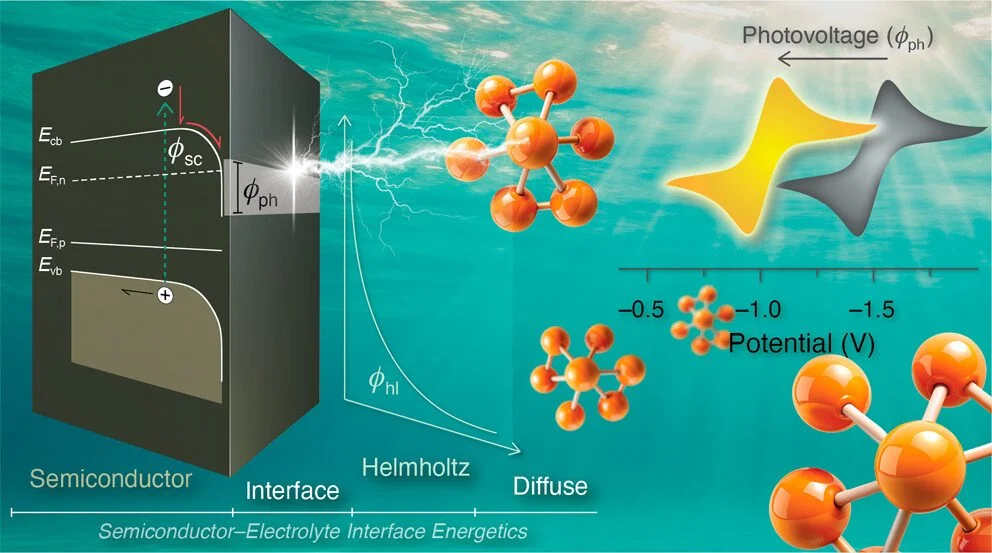
Small changes in the chemical potential at a semiconductor interface can result in dramatic changes to the space-charge layer that underpins applications in the electronic and photovoltaic industries as well as in photoelectrochemical cells for fuel production. There has hence been great interest in techniques that directly probe the space-charge layer, yet many fail at the semiconductor–electrolyte interface due to the potential drop in the electric double-layer region of the electrolyte. This article demonstrates that photovoltages, obtained from straightforward cyclic voltammetry measurements, provide an experimental and quantitative approach for characterizing the semiconductor–electrolyte interface. Key parameters accessible through this approach include the flat-band potential (Efb), the fraction of the total potential that drops across the space-charge layer (γsc) and the electric double layer, as well as the surface recombination lifetime (τs). We report photovoltage measurements for p-type Si(111) photoelectrodes in contact with electrolytes containing redox-active species with a range of known reduction potentials that exceed the 1.1 eV bandgap. In tetrabutylammonium [NBu4]+ electrolyte, the flat-band potential determined for hydrogen-terminated (p-Si–H), methyl-terminated (p-Si–CH3), and chemically oxidized (p-Si–cSiOx) surfaces were −0.02, −0.31, and 0.30 V vs Fc+/0, respectively, agreeing well with expected shifts arising from surface dipole modifications. The quantitative analysis also reveals that 67% of the applied bias drops across the space-charge layer for p-Si–H, 73% for p-Si–CH3, and only 44% for p-Si–cSiOx. The remaining potential drop is attributed to the interfacial surface layer, which consists of a molecular dipole or oxide overlayer, and the Helmholtz layer within the electrolyte. When the larger [NBu4]+ electrolyte was replaced with Li+, the flat-band position showed minimal changes, but the fraction of the potential drop across the space-charge layer increased significantly, consistent with the small cation altering the structure of the electric double layer.
Vecchi, P.; Goodwin, M. J.; Leimkuhl, D. P.; Dickenson, J. C.; Bein, G. P.; Jackson, M. N.; Cahoon, J. F.; Dempsey, J. L.; Meyer, G. J.; Sampaio, R. N. Quantitative Analysis of the Semiconductor–Electrolyte Interface Using Cyclic Voltammetry Measurements. J. Am. Chem. Soc. 2026, 148 (10), 10491–10505. https://doi.org/10.1021/jacs.5c18135